Crude oil powers much of the modern world, fueling transportation, heating homes, and serving as a base for countless industrial products. Yet despite its widespread use, crude oil is classified as a non-renewable resource. This means that once it's extracted and consumed, it cannot be replenished within a human timescale. Understanding why crude oil is non-renewable is essential for making informed decisions about energy policy, environmental protection, and sustainable development.
The classification isn’t arbitrary—it’s grounded in geology, chemistry, and time. Unlike solar or wind energy, which are continuously available, crude oil forms over millions of years under specific conditions that cannot be replicated quickly. As global demand continues to rise, the finite nature of this resource becomes increasingly critical.
How Crude Oil Forms: A Geological Timeframe
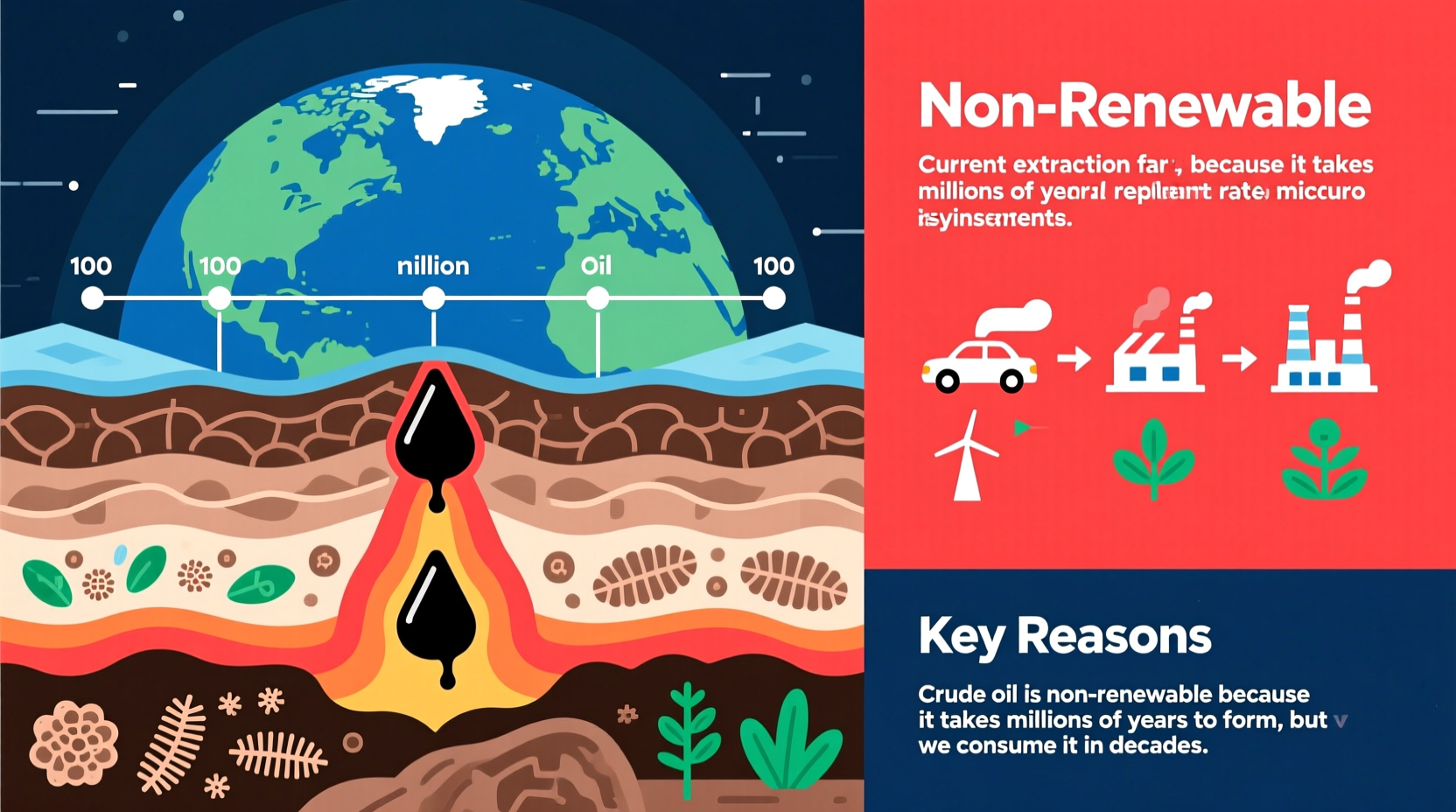
Crude oil originates from ancient organic matter—primarily marine microorganisms like algae and plankton—that lived millions of years ago. When these organisms died, they sank to the bottom of oceans and lakes, where they were buried under layers of sediment. Over time, heat and pressure transformed this organic material into kerogen, and eventually into hydrocarbons—oil and natural gas.
This transformation process takes place deep underground, typically between 2 to 5 kilometers below the surface, where temperatures range from 90°C to 160°C (194°F to 302°F). These conditions must remain stable for tens of millions of years. The entire formation cycle—from biological deposition to usable crude oil—can span anywhere from 10 million to over 100 million years.
“Oil doesn’t form overnight. It requires precise geological conditions and immense spans of time—far beyond human lifespans or even civilizations.” — Dr. Alan Reyes, Geoscientist at the Energy Research Institute
Because new oil deposits aren’t forming at a rate anywhere close to current consumption levels, the supply is effectively fixed. Even if microscopic life continues to die and settle today, it would take tens of millions of years before any meaningful quantity of new oil could be generated—making crude oil functionally non-renewable.
Rate of Consumption vs. Rate of Formation
One of the most compelling reasons crude oil is non-renewable lies in the staggering imbalance between how fast we use it and how slowly it forms. According to the U.S. Energy Information Administration (EIA), the world consumes approximately 100 million barrels of oil per day. That equates to roughly 37 billion barrels annually.
In contrast, the natural processes that create oil operate on geological timescales. To replace just one barrel of oil through natural means would require thousands to millions of years—depending on subsurface conditions. There is no known mechanism by which this gap can be closed using natural systems alone.
Global Oil Reserves and Depletion Timeline
As of 2024, proven global oil reserves stand at approximately 1.7 trillion barrels. At current consumption rates, this suggests around 50 years of supply remains. However, this estimate assumes no increase in demand, no geopolitical disruptions, and full access to all known reserves—all optimistic assumptions.
Moreover, not all reserves are equally accessible or economically viable to extract. As easily reachable oil fields deplete, companies turn to more complex sources like deepwater drilling, tar sands, and shale oil, which come with higher costs and greater environmental risks.
Environmental and Chemical Constraints
Beyond time and volume, there are fundamental environmental limitations preventing oil from being renewable. For oil to form, several strict conditions must align:
- Dense accumulation of organic material in low-oxygen environments
- Deep burial beneath impermeable rock layers to prevent escape
- Consistent heat and pressure over millions of years
- Presence of source rock capable of generating hydrocarbons
These conditions existed during certain periods in Earth’s history—such as the Jurassic and Cretaceous eras—but are rare today. Modern oceanic sediments contain far less organic concentration, and many regions lack the tectonic activity needed to generate the necessary heat and pressure.
Additionally, when oil is burned, its carbon content is released into the atmosphere as CO₂—a greenhouse gas. This chemical transformation is irreversible on short timescales. While plants and oceans absorb some of this carbon, they do so slowly, and the balance has been disrupted by rapid fossil fuel combustion since the Industrial Revolution.
Why Recycling Isn’t Possible
Unlike materials such as glass, metal, or paper, crude oil cannot be recycled after use. Once refined and combusted in engines or power plants, the hydrocarbons break down into water vapor and carbon dioxide. These byproducts cannot be reassembled into usable oil through any practical or scalable method.
Some experimental technologies aim to capture CO₂ and convert it back into synthetic fuels using renewable energy, but these processes are energy-intensive, expensive, and still in early stages. They do not constitute true recycling of oil but rather alternative fuel synthesis—and even then, they don't restore the original crude deposit.
This irreversibility underscores the non-renewable nature of oil: every drop burned is permanently lost from the usable pool.
Comparison of Energy Sources: Renewable vs. Non-Renewable
| Energy Source | Renewable? | Replenishment Time | Environmental Impact |
|---|---|---|---|
| Crude Oil | No | Millions of years | High (CO₂ emissions, spills) |
| Solar Power | Yes | Instantaneous (daily) | Low (manufacturing impact only) |
| Wind Energy | Yes | Ongoing | Very low |
| Coal | No | Over 100 million years | Very high |
| Biomass | Yes (if sustainably managed) | Months to decades | Moderate (land use, emissions) |
Real-World Example: The North Sea Oil Decline
A clear illustration of oil’s non-renewability can be seen in the North Sea oil fields, once among the most productive in Europe. Discovered in the late 1960s, these offshore reserves fueled the UK and Norway for decades. Production peaked in the late 1990s and has been in steady decline ever since.
Despite advances in extraction technology, including enhanced recovery methods, output continues to fall because the reservoirs are physically depleted. No amount of investment or innovation can restore the original volume of oil. New exploration in the region yields smaller finds, requiring more effort for less return.
This case exemplifies a broader trend: once an oil field passes its peak production, decline is inevitable. There is no “renewal” phase—only diminishing returns until extraction ceases altogether.
Actionable Steps Toward Sustainable Alternatives
Recognizing crude oil as non-renewable compels individuals, industries, and governments to act. Here is a checklist of practical steps to reduce dependence on finite fossil fuels:
Checklist: Reducing Reliance on Crude Oil
- Switch to electric vehicles or public transportation where possible
- Support policies promoting renewable energy infrastructure
- Improve home insulation to reduce heating oil consumption
- Advocate for corporate sustainability reporting and clean energy adoption
- Educate others on the long-term risks of fossil fuel dependency
- Invest in green technologies or funds focused on sustainability
Frequently Asked Questions
Can crude oil be made artificially to replace natural supplies?
While synthetic fuels can be created using processes like Fischer-Tropsch synthesis, they require significant energy input and raw materials such as coal or captured CO₂. These are not replacements for naturally occurring crude oil and are not scalable enough to offset depletion.
If oil comes from dead plants and animals, why can’t we just grow more to make new oil?
The issue is time and conditions. Today’s organic waste decomposes aerobically or is processed too quickly to form oil. The unique anaerobic, high-pressure environments required no longer exist widely, and even if they did, the process would still take millions of years.
Will we run out of oil completely?
We may never \"run out\" in the sense of zero remaining, but economically viable and easily accessible oil will become increasingly scarce. Future supplies will likely come from costly, environmentally risky sources, leading to higher prices and reduced usability.
Conclusion: A Call for Informed Action
Crude oil is non-renewable because its formation spans millions of years, its consumption occurs in seconds, and its chemical transformation upon use is irreversible. These factors combine to make oil a finite resource—one we are using far faster than nature can replenish.
The implications extend beyond energy security to climate change, economic stability, and intergenerational equity. Transitioning to renewable alternatives isn’t just an environmental ideal; it’s a necessity driven by basic physical reality.









 浙公网安备
33010002000092号
浙公网安备
33010002000092号 浙B2-20120091-4
浙B2-20120091-4
Comments
No comments yet. Why don't you start the discussion?