Xolair (omalizumab) is a groundbreaking biologic medication used to treat moderate-to-severe allergic asthma, chronic idiopathic urticaria (CIU), and nasal polyps. Despite its proven efficacy in reducing symptoms and improving quality of life, many patients are shocked by its price—often exceeding $1,000 per dose and reaching tens of thousands annually. This raises a critical question: Why is Xolair so expensive? The answer lies in a complex interplay of scientific innovation, regulatory hurdles, production challenges, and healthcare system dynamics.
Understanding the cost of Xolair requires looking beyond the pharmacy counter. It involves examining the entire lifecycle of a specialty biologic—from initial research to patient access. While the high price tag can feel unjustified, it reflects real financial and logistical investments. At the same time, systemic inefficiencies and market exclusivity play a role. This article breaks down the factors driving Xolair’s cost and offers practical guidance for managing expenses.
The Science Behind Xolair: A Biologic, Not a Simple Pill
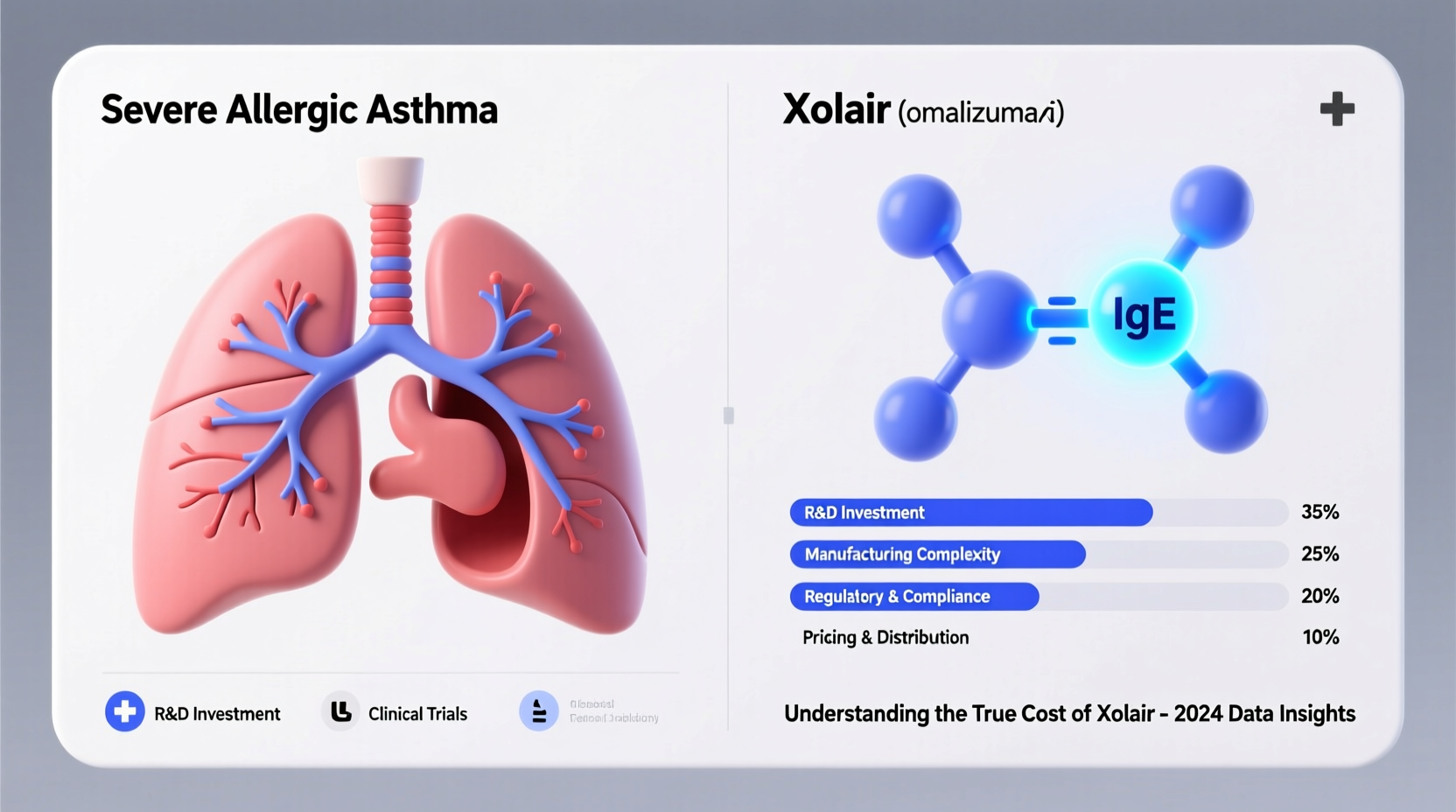
Unlike traditional small-molecule drugs synthesized from chemical compounds, Xolair is a monoclonal antibody—a type of biologic drug produced using living cells. This distinction is crucial. Biologics like Xolair are large, complex proteins engineered to interact precisely with the immune system. Specifically, Xolair targets immunoglobulin E (IgE), a key player in allergic reactions, preventing it from triggering inflammation.
Manufacturing such a molecule is vastly more complicated than producing aspirin or antihistamines. It requires genetically modified cell lines (typically Chinese hamster ovary cells), sterile bioreactors, and multi-step purification processes under strict quality control. Even minor changes in temperature or pH during production can alter the protein’s structure and render it ineffective—or worse, unsafe.
“Biologics represent the frontier of precision medicine, but their complexity comes at a steep cost. You’re not just paying for a drug; you’re paying for years of molecular engineering and ultra-precise manufacturing.” — Dr. Linda Park, Immunology Researcher, Stanford University
Research and Development: The Hidden Cost of Innovation
The journey from concept to FDA approval for Xolair spanned over a decade and involved hundreds of clinical trials. Genentech and Novartis, the companies behind Xolair, invested heavily in identifying IgE as a therapeutic target, designing the antibody, and testing its safety and efficacy across diverse patient populations.
Industry estimates suggest that bringing a single biologic drug to market costs between $1 billion and $2.6 billion when factoring in failed candidates, regulatory compliance, and opportunity cost. Only a fraction of experimental drugs succeed. For every approved therapy like Xolair, dozens of others fail in early stages, with no return on investment.
Xolair was first approved by the FDA in 2003 for asthma and later expanded to CIU and nasal polyps. Each new indication required additional trials, adding to development costs. Because the drug remains under patent protection and faces no direct biosimilar competition in the U.S., the manufacturers maintain pricing power to recoup R&D investments.
Manufacturing and Supply Chain Challenges
Producing Xolair isn’t scalable like mass-producing generic tablets. Each batch must be individually validated, tested for potency, and stored under refrigeration. The cold chain logistics—from factory to clinic—add significant overhead. Any break in temperature control risks compromising the drug’s integrity, leading to waste and increased costs.
Moreover, Xolair is dosed based on body weight and baseline IgE levels, meaning supply must accommodate variable treatment regimens. Unlike fixed-dose medications, this customization complicates inventory planning and increases per-patient variability in cost.
Insurance, Reimbursement, and Patient Access Barriers
Even with insurance, patients often face high out-of-pocket costs due to Xolair’s classification as a “specialty drug.” Many health plans place it on the highest tier, requiring prior authorization and step therapy—meaning patients must try cheaper treatments first before gaining coverage.
Medicare Part B covers Xolair when administered in a clinical setting, but Part D plans may impose deductibles and coinsurance. For self-insured individuals or those with high-deductible plans, monthly costs can exceed $1,500 before meeting their deductible.
| Insurance Type | Coverage Model | Typical Patient Responsibility |
|---|---|---|
| Medicare Part B | Administered in-office | 20% coinsurance after deductible |
| Commercial Insurance | Prior auth + tier 4 copay | $100–$500 per injection |
| Medicaid | Varies by state | Low or no cost |
| Uninsured | No coverage | $3,000–$5,000+ monthly |
Real-World Example: Sarah’s Struggle with Affordability
Sarah, a 34-year-old teacher from Ohio, was diagnosed with chronic hives unresponsive to antihistamines. Her dermatologist prescribed Xolair, which dramatically reduced her symptoms within weeks. But her insurance required prior authorization and denied the first two requests, citing lack of trial with alternative therapies. After three months of appeals and escalating medical bills, she was finally approved—but still faced a $300 copay per monthly injection.
“I felt trapped,” Sarah said. “The drug gave me my life back, but I had to choose between paying for it and covering rent. It shouldn’t be this hard to get a life-changing medication.”
Strategies to Reduce Out-of-Pocket Costs
While Xolair’s list price is non-negotiable, several pathways exist to reduce what patients actually pay. Proactive engagement with support programs and healthcare providers is essential.
Step-by-Step Guide to Lowering Xolair Costs
- Contact the Xolair Co-pay Program: Visit xolair.com and enroll in the XOLAIR® Mobile App & Savings Card. Eligible commercially insured patients may pay as little as $0 per dose, up to a maximum annual benefit.
- Apply for Patient Assistance: Uninsured or low-income patients can apply through Genentech Access Solutions for free medication.
- Verify Administration Setting: If possible, receive injections in a physician’s office billed under Medicare Part B rather than outpatient pharmacy (Part D), where reimbursement structures favor lower patient costs.
- Appeal Insurance Denials: Work with your doctor to submit detailed letters of medical necessity. Persistence often leads to approval.
- Explore Foundation Support: Organizations like PAN Foundation and HealthWell Foundation offer grants for asthma and autoimmune conditions.
Frequently Asked Questions
Will a generic version of Xolair ever be available?
Not exactly. Because Xolair is a biologic, it cannot have a “generic” equivalent. However, biosimilars—highly similar versions—are in development. The first biosimilar to omalizumab received FDA approval in 2024 and is expected to launch in 2025, potentially cutting prices by 20–40%.
Why doesn’t Medicare cover the full cost?
Medicare Part B covers 80% of the approved amount after the deductible. Patients are responsible for the remaining 20%, unless they have supplemental insurance (Medigap) or qualify for Extra Help.
Can I buy Xolair internationally to save money?
Some patients consider purchasing from Canada or Europe, where prices are lower due to government negotiation. However, importing prescription drugs is illegal in the U.S. and poses safety risks due to counterfeit products and improper storage.
Conclusion: Navigating Cost with Knowledge and Advocacy
Xolair’s high price reflects genuine scientific achievement and operational complexity, but also highlights flaws in the U.S. pharmaceutical pricing system. While innovation deserves reward, patients should not bear unsustainable burdens for life-altering treatments. Understanding the drivers of cost empowers individuals to seek financial aid, challenge denials, and advocate for change.
With the arrival of biosimilars on the horizon and growing public pressure for drug pricing reform, there is hope for more affordable access in the coming years. Until then, take action: explore co-pay cards, engage patient assistance programs, and work closely with your care team. Your health matters—and so does your financial well-being.









 浙公网安备
33010002000092号
浙公网安备
33010002000092号 浙B2-20120091-4
浙B2-20120091-4
Comments
No comments yet. Why don't you start the discussion?